Reference
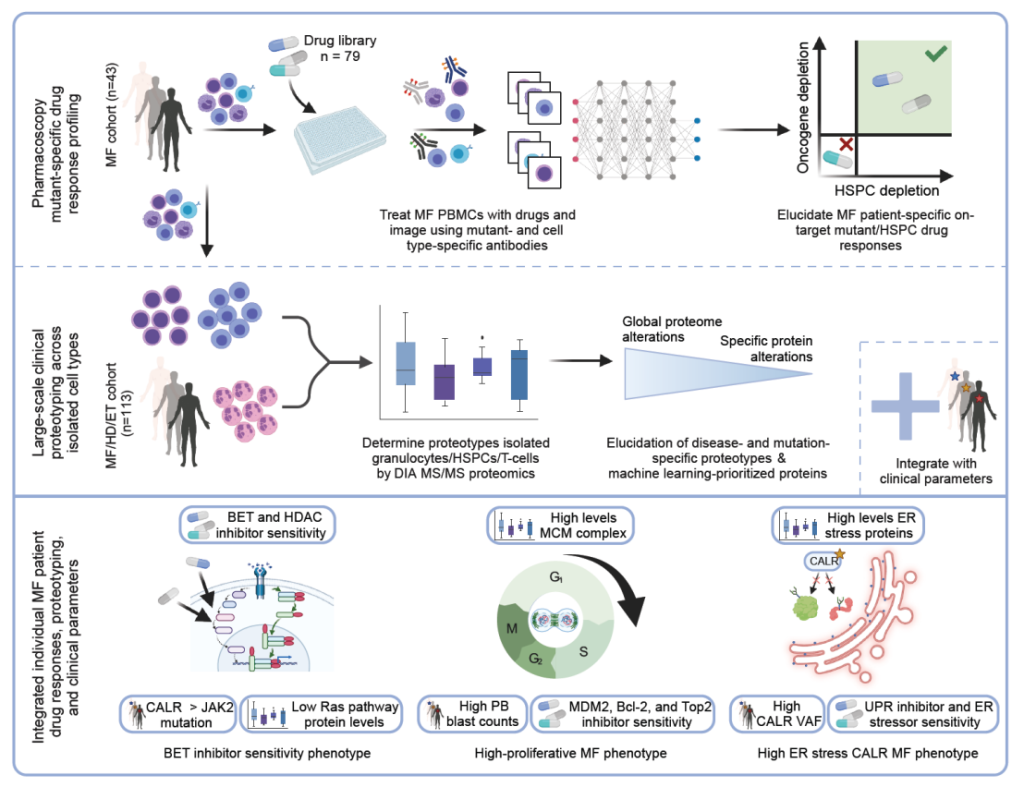
Wildschut, M.H.E., Mena, J., Dördelmann, C. et al. Proteogenetic drug response profiling elucidates targetable vulnerabilities of myelofibrosis. Nat Commun 14, 6414 (2023). https://doi.org/10.1038/s41467-023-42101-z
- Prof. Dr. Berend Snijder
ETH Zürich
- iDoc